Mosquitoes carrying malaria are evolving more quickly than insecticides can kill them – researchers
Once-lethal insecticides now require concerningly high concentrations to effectively kill mosquitoes. Some mosquitoes are already immune to all classes of insecticides used to control malaria.

The fight against infectious disease is a race against evolution. Bacteria become resistant to antibiotics. Viruses adapt to spread more quickly. Diseases transmitted by insects present another evolutionary front: Insects themselves can evolve resistance to the poisons that people use to kill them.
In particular, the mosquito-borne disease malaria kills over 600,000 people annually. Since World War II, people have battled malaria with insecticides – chemical weapons intended to kill Anopheles mosquitoes infected with the Plasmodium parasites that cause the disease.
However, mosquitoes are quickly evolving counterstrategies that make these insecticides ineffective, putting millions of people at greater risk of deadly infection. My colleagues and I have newly published research showing how.
Insecticide resistance threatens public health
As an evolutionary geneticist, I study natural selection – the basis for adaptive evolution. Genetic variants that best promote survival can replace less advantageous versions, causing species to change. Anopheles mosquitoes are frustratingly adept at evolving.
In the mid-1990s, most African Anopheles were susceptible to pyrethroids, a popular type of insecticide originally derived from chrysanthemums. Anopheles control relies on two pyrethroid-based methods: insecticide-treated bed nets to protect sleepers, and indoor residual spraying of insecticide against the walls of homes. These two methods alone likely prevented over a half-billion cases of malaria between 2000 and 2015.
However, mosquitoes today from Ghana to Malawi are often able to survive insecticide concentrations 10 times the previously lethal dose. Along with Anopheles control efforts, agriculture also inadvertently exposes mosquitoes to pyrethroids and contributes to insecticide resistance.
In some African locales, Anopheles is already showing resistance to all four main classes of insecticide used for malaria control.
Adaptation in Latin American mosquitoes
Anopheles mosquitoes and the malaria-causing Plasmodium also occur outside Africa, where insecticide resistance is less well-researched.
In much of South America, the main malaria vector is Anopheles darlingi. This mosquito species has diverged evolutionarily from the African vectors so extensively that it might be a different genus, Nyssorhynchus. Along with colleagues from eight countries, I analyzed over 1,000 Anopheles darlingi genomes to understand its genetic diversity, including any recent changes due to human activity. My collaborators collected these mosquitoes at 16 locations ranging from the Atlantic coast of Brazil to the Pacific side of the Andes in Colombia.
We found that, like its African counterparts, Anopheles darlingi shows extremely high genetic diversity – more than 20 times that of humans – indicating that very large populations of this insect exist. A species with such a vast gene pool is well poised to adapt to new challenges. The right mutation giving it the advantage it needs is more likely to pop up when there are so many individuals. And once that mutation starts to spread, it’s protected by numbers since it won’t be wiped out if a few mosquitoes die by chance.
In contrast, bald eagles in the contiguous U.S. were never able to evolve resistance against the insecticide DDT and approached extinction. Evolution is more efficient among millions of insects than mere thousands of birds. And indeed, we saw signals of adaptive evolution in the resistance-related genes of Anopheles darlingi occurring over the past few decades.
Mosquitoes evolve to detoxify poisons
Insecticides like pyrethroids and DDT share the same molecular target: channels in nerve cells that can open and close. When open, the nerve cell stimulates other cells. These insecticides force the channels to remain open and continuously fire, causing paralysis and death. However, insects can evolve resistance by changing the shape of the channel itself.
Earlier genetic scans performed by other researchers had not detected this type of resistance in Anopheles darlingi, and neither did ours. Instead, we found that resistance is evolving in another way: a group of genes encoding enzymes that break down toxic compounds. High activity of these enzymes, called P450, frequently underlies resistance to insecticides in other mosquitoes. The same cluster of P450 genes has changed independently at least seven times across South America since insecticide use began in the mid-20th century.
In French Guiana, a different set of P450 genes exhibits a similar evolutionary pattern, cementing the clear connection between these enzymes and adaptation. Moreover, when we exposed mosquitoes to pyrethroids in sealed bottles, differences among the P450 genes of individual mosquitoes were linked to the length of time they stayed alive.
Insecticide-heavy campaigns against malaria have been only sporadic in South America and may not be the main driver behind this evolution. Instead, it’s possible that mosquitoes are being exposed indirectly to agricultural insecticides. Intriguingly, we saw the strongest signs of evolution in places where farming is prevalent.
Toward more sophisticated vector control
Despite new vaccines and other recent advances against malaria, mosquito control remains essential for reducing disease.
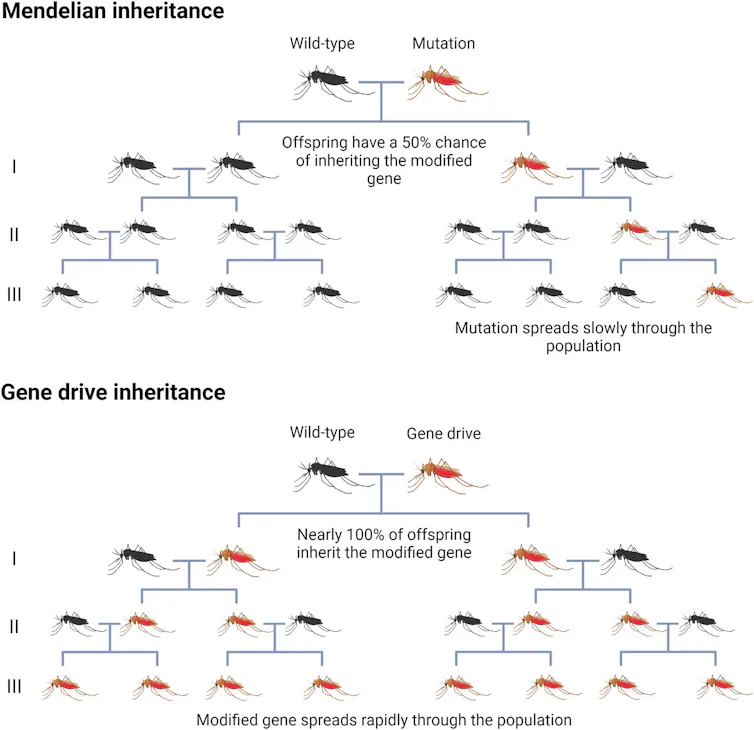
Some countries are launching trials of gene drives to control malaria, which involve forcing a genetic modification into a mosquito population to reduce their numbers or their tolerance for Plasmodium. Such prospects are exciting, though the relentless adaptability of mosquitoes could be an obstacle.
I and others are revising methods to efficiently test for emerging insecticide resistance. Genome-scale sequencing remains important to detect new or unexpected evolutionary responses. The risk of adaptation is highest under a continuous, strong selection pressure, so minimizing, switching and staggering pesticides can help thwart resistance.
Success in the fight against evolving resistance will require a coordinated effort of monitoring, and reacting accordingly. Unlike evolution, humans can think ahead.
Jacob A Tennessen receives funding from the National Institutes of Health via Harvard T.H. Chan School of Public Health and the Broad Institute.
Read These Next
For 2 centuries, Latter-day Saints have revered religious freedom – but their definition is evolving
Latter-day Saints have long valued the US Constitution’s promise of religious freedom – but the…
Getting disability benefits got harder after the Social Security Administration’s staff was slashed
Researchers learned from dozens of interviews that the usual ways of resolving complex cases, escalating…
Your phone screen doesn’t have the same color range as the human eye – and AI widens the gap between
A photographer describes how your phone camera flattens and dulls the colors in photos – and how to…




