Fat cells burn energy to make heat – making them the next frontier of weight loss therapies
GLP-1 drugs like Ozempic and Wegovy aid in weight loss by suppressing appetite. Researchers are looking next at how the body expends energy – including through fat.

Over the past few years, a new class of medications has transformed the treatment of obesity. Drugs like Ozempic, Wegovy and Mounjaro work primarily by reducing appetite, helping people eat less and feel full sooner. Their success has demonstrated something important: Body weight is biologically regulated, and targeting the right biological pathways can lead to meaningful weight loss that can help transform lives.
But appetite is only half of the equation. Your weight reflects a balance between the calories you consume through your diet and the energy you expend through movement, exercise and maintaining basic cellular function. While recent therapies have focused on controlling energy intake, scientists are increasingly turning their attention to the other side of the ledger: the tissues that burn energy.
At the center of this conversation is an organ most people misunderstand: fat. For decades, fat – also known as adipose tissue – was thought of as passive storage: a biological pantry for excess calories. Scientists now know this view is incomplete.
Fat is not just storage
White adipose tissue, the most abundant type of fat in adults, does store energy in the form of triglycerides. But it also has several other functions.
For one, white fat is a powerful endocrine organ, releasing hormones like leptin that reduce appetite, as well as adiponectin, which regulates insulin and blood sugar levels. It also cushions organs, insulates against heat loss and acts as a metabolic buffer, safely storing excess lipids that would otherwise accumulate in the liver or muscle.
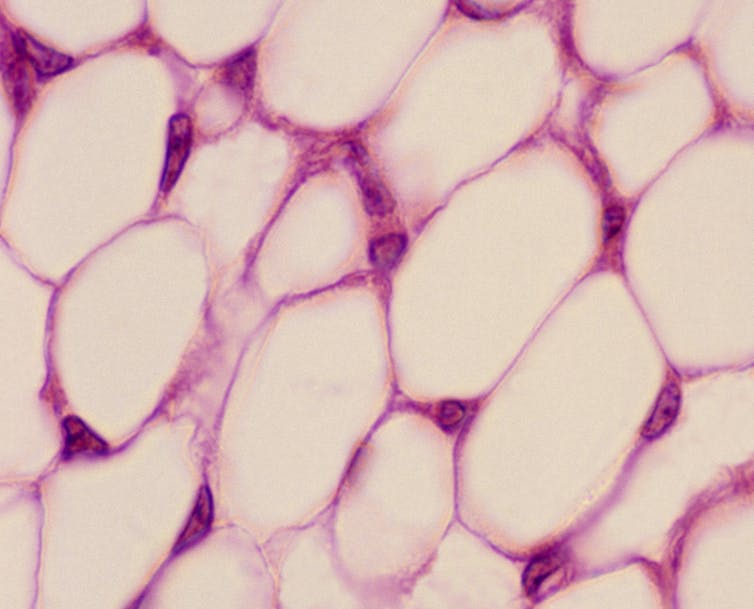
When white adipose cells expand in a healthy, flexible way, they protect the body. When they become inflamed or dysfunctional, they contribute to insulin resistance, fatty liver disease and cardiovascular risk. Obesity arises from both the expansion of white adipose cells and an increase in their number.
In other words, fat is not inherently harmful. Its health impact depends on the size of adipose cells, and when they become too large, they are unable to function optimally. Increasing the number of new fat cells can sometimes improve metabolic function.
Moreover, there are additional types of fat, and they behave in different ways.
Brown fat: The cellular furnace
Unlike white fat, brown fat is specialized to burn energy. Brown adipose cells are packed with mitochondria – the tiny power plants inside cells – and contain a protein called UCP1 that allows them to convert chemical energy directly into heat. Instead of storing calories, brown fat dissipates them.
In infants, brown fat helps maintain body temperature. For years, scientists believed it largely disappeared in adulthood. But imaging studies in the late 2000s revealed that many adults retain metabolically active brown fat, particularly in the neck and upper chest.
Exposure to cold temperatures naturally triggers the brain to stimulate brown fat cells and generate heat. As energy use rises for this process, so does calorie-burning.
If activating brown fat increases energy expenditure, could it be harnessed to treat obesity?
The challenge is that human metabolism is tightly regulated. When energy expenditure increases, the body often compensates by stimulating hunger. Studies in animals – and observations in humans – show that cold exposure not only activates brown fat but also increases appetite. The brain detects the higher energy demand and signals for greater food intake.
From an evolutionary perspective, this makes sense. For our human ancestors, cold environments meant survival required more fuel. A system that failed to replace calories burned to keep you warm would have been dangerous. This homeostatic defense of body weight is powerful. It is one reason why weight loss is difficult to sustain and why increasing energy expenditure alone may not be sufficient to lose weight.
But when coupled with GLP-1 drugs that suppress appetite, promoting energy expenditure could lead to therapies that are even more powerful at promoting weight loss.

Beige fat and metabolic plasticity
Adding further complexity to fat’s role in weight loss are beige fat cells. These cells arise within white fat depots under certain conditions – such as cold exposure or specific hormonal signals – and acquires some of the heat-producing properties of brown fat. This process, often called browning, reveals that adipose tissue is remarkably flexible.
Fat is not a static mass. It contains stem and progenitor cells capable of generating new adipocytes with distinct properties. That flexibility opens intriguing therapeutic possibilities: Instead of merely shrinking fat, could researchers reprogram it to become something else?
Researchers like me are exploring ways to safely enhance the heat-generating capacity of fat cells, potentially increasing energy expenditure without relying solely on environmental cold. Brown and beige fat are compelling targets because they are purpose-built for heat production, which is why my lab is focusing on harnessing them to treat metabolic disease.
But fat is not the only tissue in the body that consumes energy or can generate heat in the cold. Skeletal muscle accounts for a substantial portion of daily energy expenditure, particularly during activity. The liver continuously engages in metabolically expensive processes. Even subtle futile cycles – processes in which molecules are repeatedly built and broken down – consume energy and generate heat.
The future of metabolic therapeutics for weight loss may involve carefully increasing energy flux across multiple tissues. The challenge is doing so without triggering compensatory hunger or unintended side effects. Any intervention that dramatically raises metabolic demand risks being interpreted by the brain as a threat to survival.

A two-sided strategy to maximize weight loss
The success of GLP-1–based medications has demonstrated that targeting appetite pathways can overcome some of the body’s resistance to weight loss. The next generation of therapies may build on that foundation.
One possibility is combining medications that modulate appetite with interventions that enhance energy expenditure. By influencing both sides of the energy balance equation – intake and output – it might be possible to achieve more durable metabolic improvements.
Equally important is shifting the public narrative. Fat is not merely an enemy to eliminate. It is a dynamic, multifunctional organ that protects, communicates, adapts and, under the right conditions, burns energy.
Understanding that complexity moves society beyond simplistic views of weight regulation. It also points toward a future in which therapies are not just about eating less, but about strategically harnessing the body’s own metabolic machinery.
The era of appetite control has begun. I believe the era of precision energy expenditure will be next.
Claudio Villanueva receives funding from National Institutes of Health.
Read These Next
Who are hospital ethics consultants, and why should you care?
Hospitals rely on ethics consultants to help navigate difficult decisions about life support, consent…
What happens to debt when someone dies?
Whether or not there’s a will, the results are the same.
Getting disability benefits got harder after the Social Security Administration’s staff was slashed
Researchers learned from dozens of interviews that the usual ways of resolving complex cases, escalating…